by Chris Hitchcock | Oct 25, 2011 | Health Care, New Research, Pharmaceutical
Prevalence of Hot Flushes and Night Sweats in UK women 54-65 In a new, large (over 10 000 women) survey of UK women aged 54-65, Myra Hunter and colleagues reported on the proportion of women who have hot flushes and night sweats (HF/NS), and on how frequent and...
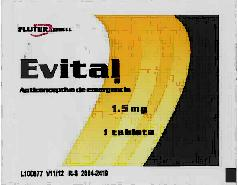
by Elizabeth Kissling | Jul 29, 2011 | Birth Control, Law/Legal
The U.S. Food and Drug Administration has issued a warning to consumers not to use the emergency birth control medicine labeled as Evital. The drug is not safe or effective in preventing pregnancy. The packaging label of the potentially ineffective and suspect...
by Elizabeth Kissling | Dec 29, 2010 | Activism, Pharmaceutical
Guest post by Leslie Botha, S.A.N.E. Vax Increasing Number of Consumers are Concerned over HPV Vaccine Safety The FDA’s December 22, 2010 ruling to expand the use of Gardasil for anal cancer prevention is unacceptable, according to Norma Erickson, President of S.A.N.E...

by Elizabeth Kissling | May 25, 2010 | Activism, Birth Control, Books, Pharmaceutical
Only a latter-day Rip Van Winkle could avoid knowing that this month marks the 50th anniversary of the FDA’s approval of Enovid, the world’s first birth control pill. Hundreds of newspaper and magazine articles have marked this anniversary. Many incorrectly credit the...

by Elizabeth Kissling | Nov 14, 2009 | Menorrhagia, Menstruation, Pharmaceutical
The FDA announced yesterday that they have approved tranexamic acid tablets for the treatment of heavy menstrual bleeding, under the brand name Lysteda. According to the press release, approximately 3 million US women experience heavy bleeding, usually with no...




